 Chemists had long known that certain elements seemed to be related to each other – they had such similar properties that they were thought of as “chemical families.” What Döbereiner found was a curious mathematical pattern among some of these families: groups of three elements in which the atomic weight of the middle element was the average of the other two. The first hints of a hidden order actually dated back half a century to a German chemist named Johann Döbereiner. Mendeleev was not the first to seek order among the elements. Hints of a Hidden Order Among the Elements By Monday evening, he had created a chart that would be the precursor of all the Periodic Tables that followed. He arranged the elements in rows of increasing atomic weight but also kept their chemical properties in mind, skipping a spot or reversing two elements if necessary to make sure that elements with similar chemical properties fell into columns. He had completed the first volume and the first two chapters of the second when he came to an impasse: He needed a way to organize the rest of the textbook, and to do that he felt he needed to organize the chemical elements themselves.Īs he grappled with this challenge over that fateful weekend, he hit upon a way to organize the 63 elements that were then known. 
Dissatisfied with the existing Russian chemistry textbooks, Mendeleev set out to write his own. Petersburg University, and one of his responsibilities was to teach introductory chemistry. What many chemistry students don’t realize is that this ubiquitous chart had its origins in the struggles of a young Russian chemistry professor over one weekend in February 1869.Īfter years of scraping together a living, Dmitri Mendeleev had finally landed a coveted post at St. 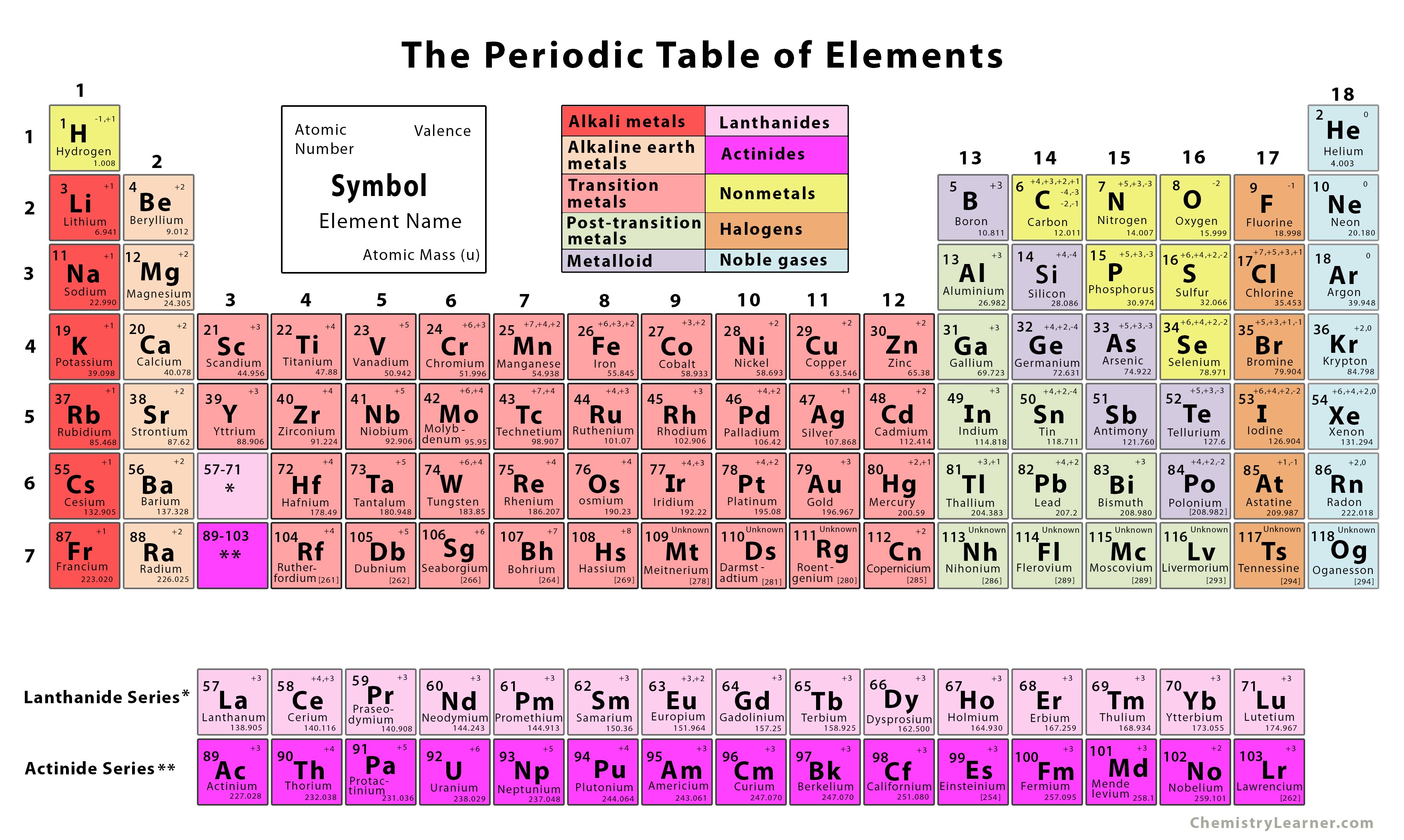
The Periodic Table of the Elements is one of the most iconic images in all of science – a fixture in every chemistry classroom in the world. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
